Neon
by Yogi P - September 9, 2023
Neon: The Colorful Gas
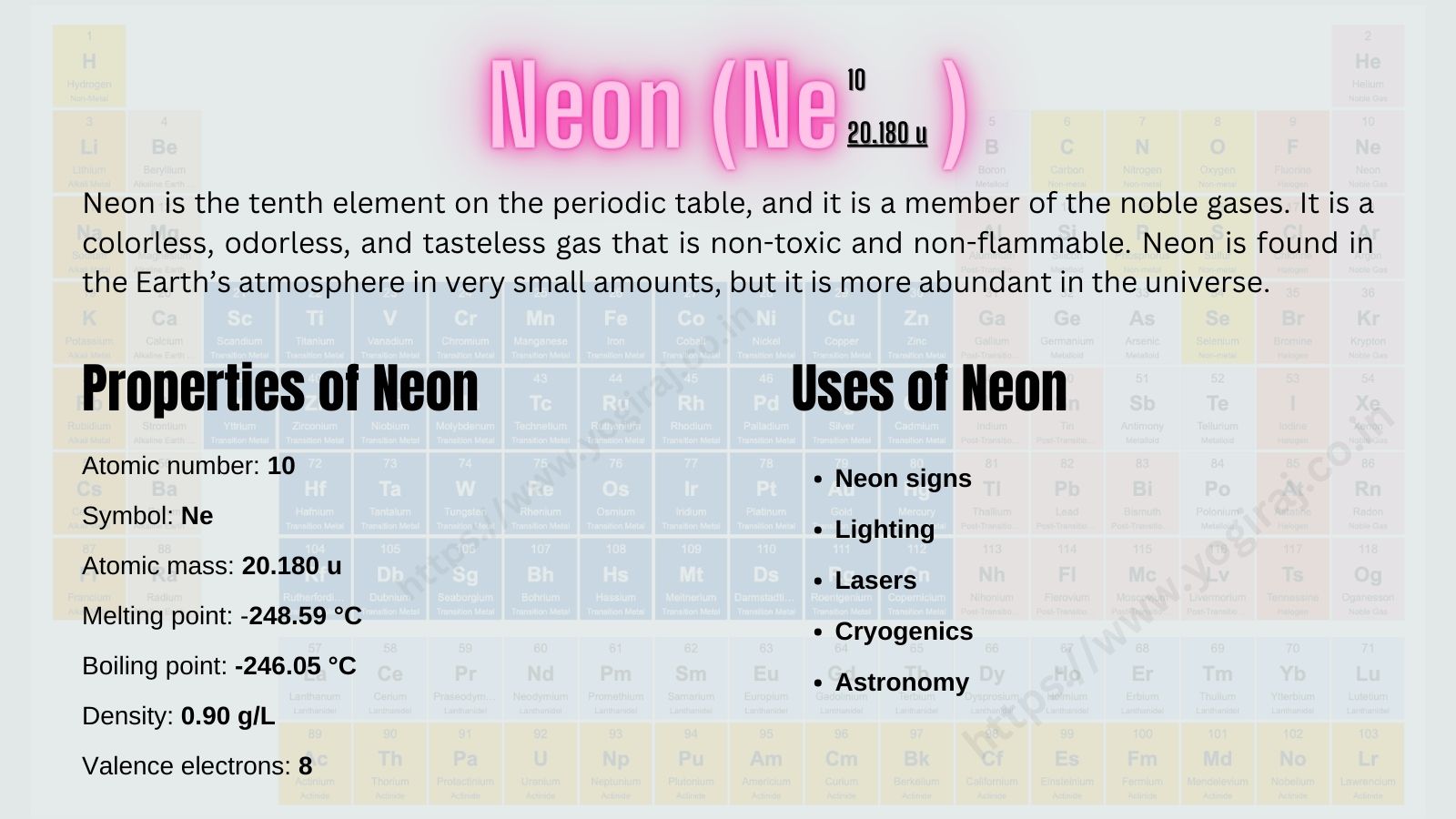
Neon is the tenth element on the periodic table, and it is a member of the noble gases. It is a colorless, odorless, and tasteless gas that is non-toxic and non-flammable. Neon is found in the Earth’s atmosphere in very small amounts, but it is more abundant in the universe.
History and Discovery of Neon
Neon was first discovered in 1898 by William Ramsay and Morris Travers. They were studying the air by fractional distillation, and they found that a small amount of gas remained that did not react with other gases. They named this gas “neon” from the Greek word “neos,” meaning “new.”
Properties of Neon
- Atomic number: 10
- Symbol: Ne
- Atomic mass: 20.180 u
- Melting point: -248.59 °C
- Boiling point: -246.05 °C
- Density: 0.90 g/L
- Valence electrons: 8
Isotopes of Neon
Neon has three naturally occurring isotopes:
- Neon-20
- Neon-21
- Neon-22
Neon-20 is the most abundant isotope, making up about 90.5% of all neon. Neon-21 is much rarer, making up about 0.27% of all neon. Neon-22 is the rarest isotope, making up about 9.23% of all neon.
Uses of Neon
Neon is used in a wide variety of applications, including:
- Neon signs: Neon signs are made by filling glass tubes with neon gas and applying a high voltage. The neon gas glows brightly, creating the characteristic colors of neon signs.
- Lighting: Neon gas is also used in lighting, such as headlights and brake lights.
- Lasers: Neon gas is used in some types of lasers.
- Cryogenics: Neon gas is used in cryogenics, the study of materials at very low temperatures.
- Astronomy: Neon gas is used in astronomy to study the composition of stars and other astronomical objects.
Safety Precautions
Neon is a safe gas, but it can be harmful if inhaled in large quantities. It can also cause frostbite if it comes into contact with skin.
Conclusion
Neon is a versatile gas that is used in a wide variety of applications. It is a safe gas, but it is important to use it with caution.






![Taxmann’s Bharatiya Criminal Laws – Bare Acts [Hindi] COMBO [Bharatiya Nyaya Sanhita (BNS) | Bharatiya Nagarik Suraksha Sanhita (BNSS) | Bharatiya Sakshya Adhiniyam (BSA)]](https://www.yogiraj.co.in/wp-content/uploads/2026/05/Taxmanns-Bharatiya-Criminal-Laws-–-Bare-Acts-Hindi.jpeg)
![Taxmann’s Criminal Laws Combo [Diglot Edition] – Bharatiya Nyaya Sanhita (BNS) | Bharatiya Nagarik Suraksha Sanhita (BNSS) | Bharatiya Sakshya Adhiniyam (BSA)](https://www.yogiraj.co.in/wp-content/uploads/2026/05/Taxmanns-Criminal-Laws-Combo.jpeg)
![Taxmann’s Bharatiya Criminal Laws – Bare Acts COMBO [Bharatiya Nyaya Sanhita (BNS) | Bharatiya Nagarik Suraksha Sanhita (BNSS) | Bharatiya Sakshya Adhiniyam (BSA)]](https://www.yogiraj.co.in/wp-content/uploads/2026/05/Bharatiya-Criminal-Laws-Bare-Acts.png)